|
Alternatively, given the near-metallic character of its lighter congener iodine, a succession of authors suggest astatine may be a metal. Chemically, studies on trace quantities of astatine, which are not necessarily reliable, have demonstrated characteristics of both metals and nonmetals. As a halogen it has usually been presumed to be a nonmetal. With no comprehensive understanding of its bulk properties, its classification remains uncertain. 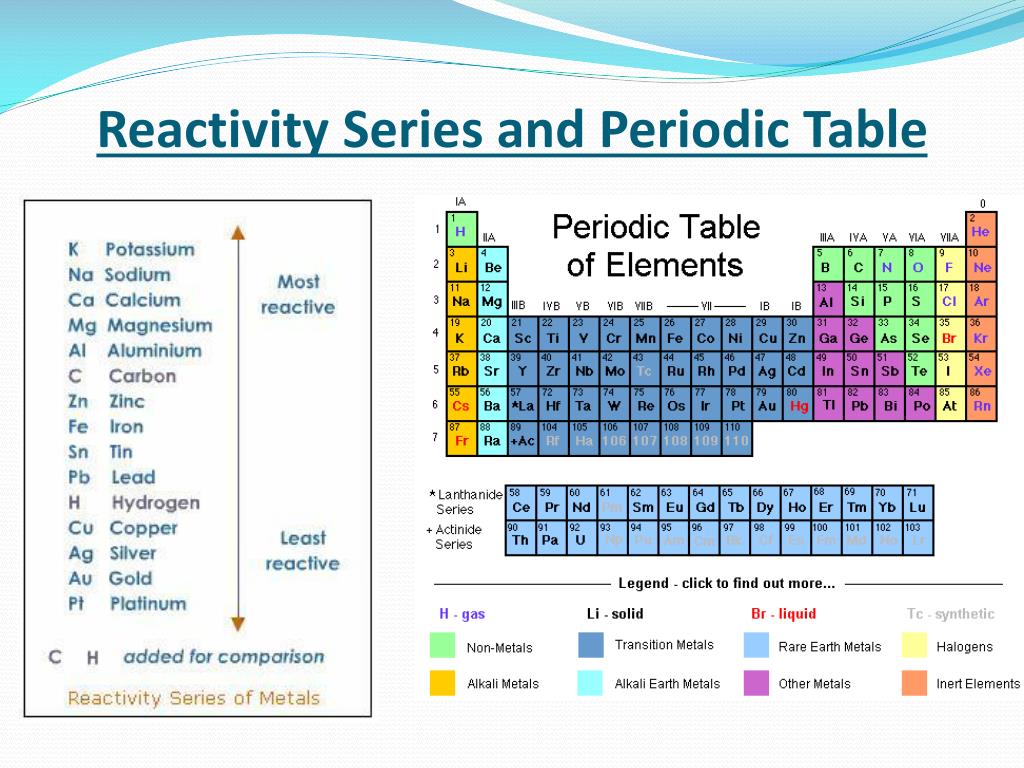
Due to its rarity and extreme radioactivity it is frequently ignored in the literature. Opinions differ as to the status of astatine. Of the 118 known elements, roughly 20% are classified as nonmetals. This article includes the metalloids for comparison. Boron, Silicon, Germanium, Arsenic, Antimony, Tellurium. 
These fourteen elements are effectively always recognized as nonmetals: Which elements are included depends on the properties regarded as most representative of nonmetallic or metallic character. There is no precise definition of a nonmetal any list of such is open to debate and revision. They lack most or all of the properties commonly associated with metals: shininess malleability and ductility good thermal and electrical conductivity and a capacity to produce basic oxides when combined with oxygen. Nonmetallic chemical elements generally have low density and high electronegativity. The chemistry of arsenic is predominately nonmetallic in nature. The vapor is lemon-yellow and smells like garlic. Since then about two dozen properties have been suggested as single criteria for distinguishing nonmetals from metals.ĭefinition and applicable elements Properties mentioned hereafter refer the most stable form of elements in ambient conditions unless otherwise mentioned Like carbon, arsenic (here sealed in a container to prevent tarnishing) vaporises rather than melts when heated. While a distinction between metals and other minerals had existed since antiquity, a basic classification of chemical elements as metallic or nonmetallic emerged only in the late 18th century. Most nonmetallic elements were not identified until the 18th and 19th centuries. Hydrogen, oxygen, carbon, and nitrogen are essential building blocks for life. They have essential uses in electronics, energy storage, agriculture, and chemical production. The distinct properties of nonmetallic elements allow for specific uses that metals often cannot achieve. Five nonmetallic elements-hydrogen, carbon, nitrogen, oxygen, and silicon-make up the overwhelming majority of the Earth's crust, atmosphere, oceans and biosphere. 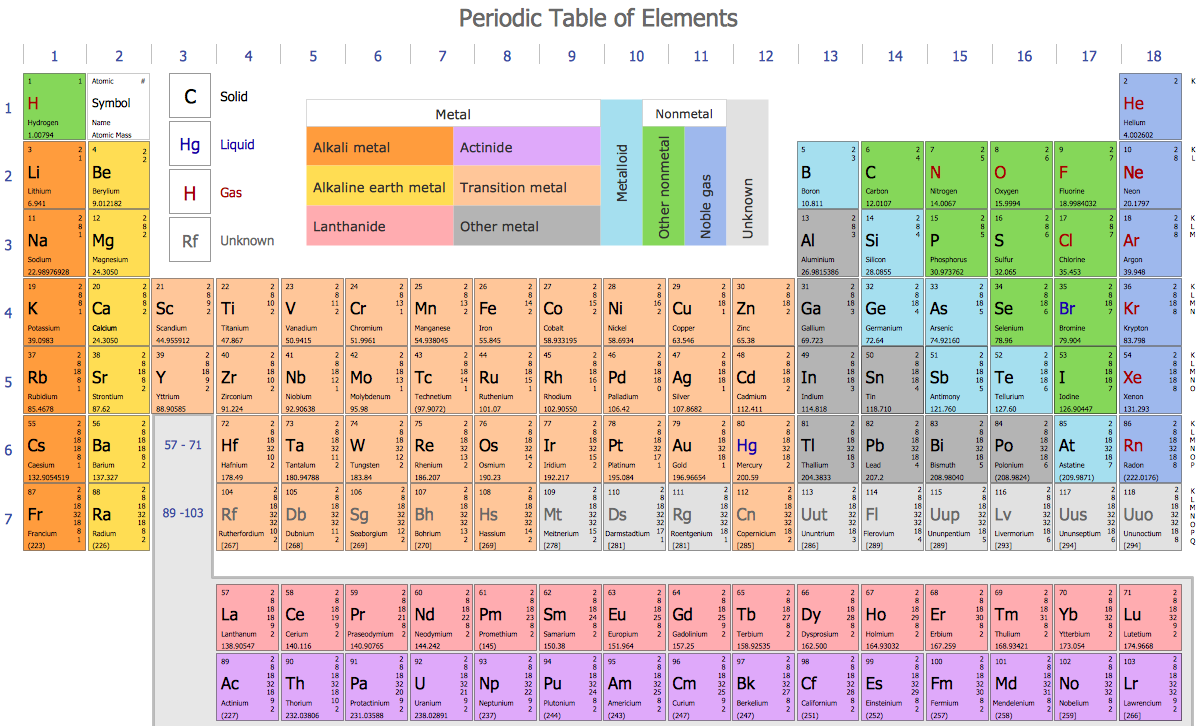
The two lightest nonmetals, hydrogen and helium, together make up about 98% of the observable ordinary matter in the universe by mass. While oxides of metals tend to be basic, those of nonmetals tend to be acidic. In contrast, metals are good conductors and most are pliable. Nonmetals are often poor conductors of heat and electricity, and tend to be brittle or crumbly as solids. They range from colorless gases like hydrogen to shiny solids like iodine. Nonmetals tend to have low density and high electronegativity (the ability of an atom in a molecule to attract electrons to itself). Complicating the situation are elements for which it becomes harder to decide whether metallic or nonmetallic properties are more pronounced. Seventeen elements are generally considered nonmetals, though some authors recognize more or fewer depending on the properties considered most representative of metallic or nonmetallic character. Ĭn Fl Og Copernicium, flerovium, and/or oganesson may turn out to be nonmetallic their status has not been confirmed.Ī nonmetal is a chemical element that mostly lacks metallic properties. 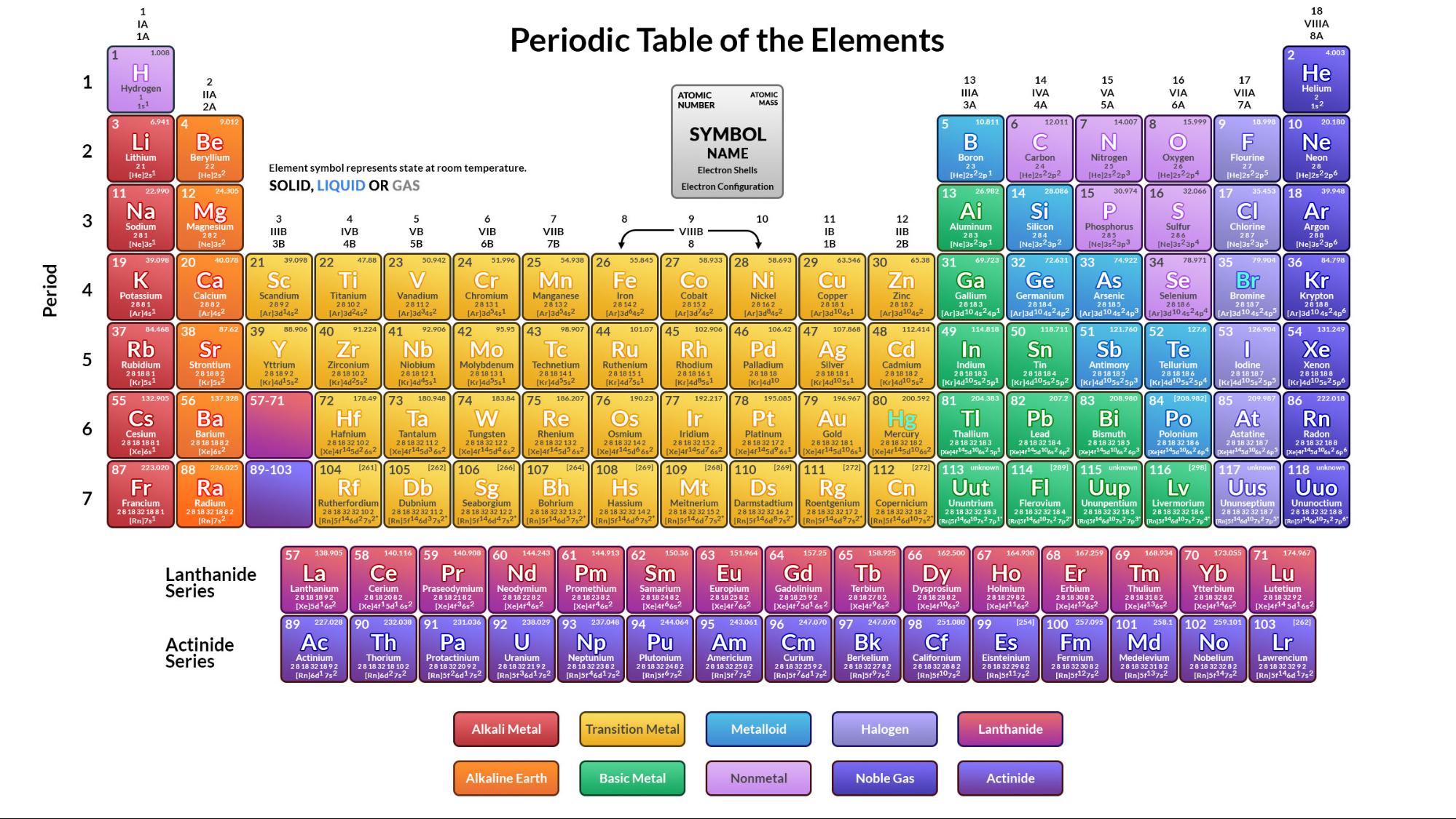
Nonmetals in their periodic table contextĪt Astatine's status is unclear while usually counted as a nonmetal, relativistic effects suggest it may be a metal. For nonmetallic substances, see materials science. For the use of the term nonmetal in astronomy, see nonmetal (astrophysics). This article is about a class of two dozen or so chemical elements.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
